Abstract
The scientific research was carried out at Lithuanian University of Health Sciences, Veterinary Academy, state enterprise “Pieno tyrimai”, as well as in dairy farms running automatic voluntary (robotic) milking systems in Lithuania. A total data set of 462574 cow milking records was assessed in the research. The objectives of this investigation were to evaluate the milk electrical conductivity indicator from robotic milking system and to assess the genetic correlation with the indices collected in a database of Lithuanian dairy cattle, to estimate heritability coefficient using multiple traits mixed linear model with permanent environment effects. The research has shown, that the electrical conductivity of milk ranged from 4.6 to 5.8 ms/cm in milk samples where somatic cell count did not exceed 200 thousand/ml and variation in electric conductivity of milk can be treated as one of the main parameters for cows’ health monitoring system. A high coefficient of heritability of electrical conductivity of milk ( = 0.512 ± 0.028; < 0.001) and a very low coefficient of heritability of somatic cell count ( = 0.032 ± 0.014; < 0.001) was determined. The results of the research have also revealed a positive genetic correlation of electrical conductivity with milk somatic cell count ( = 0.332 ± 0.016; < 0.001). Electrical conductivity of milk from robotic milking system can be introduced as an indicator of mastitis prevention in dairy cows and genetic selection based on this trait may be possible.
1. Introduction
Mastitis is usually considered one of the major diseases which leads to significant economic losses mainly due to discarded milk, decreased milk production, quality, early culling, and increased health care costs. In recent years automated milking system (AMS) robots are being introduced in dairy farms of many countries. Cows milked with the robotic milking system produced significantly more milk [1]. Research has shown that milking robots helps diagnose bovine diseases in early stages, also improve herd health and milk quality [2].
Direct selection against mastitis is difficult because of the low heritability of this trait [3]. In most countries, selection for reduced incidence of mastitis is mainly based on indicator traits such as somatic cell score and udder conformation traits. Possibly an alternative indicator trait may be electrical conductivity (EC) of milk. EC is a measure of the resistance of a particular material to an electric current. Because of increased blood capillary permeability, mastitis causes a change in ion concentrations and thus in EC [4-6]. The EC is a low cost and easy recordable information in dairy herds with automatic milking systems [7]. Electrical conductivity of milk can be detected in-line in the milking systems equipped with sensors for measuring it. Milk electrical conductivity can represent a possible indicator used for breeding programs, but there are several additional investigations needed to estimate the genetic parameters which concern the correlations of mastitis presence [6] and coefficient of heritability [8, 9].
The objectives of this investigation were to evaluate the milk electrical conductivity indicator from robotic milking system and to assess the genetic correlation with the indices collected in a database of Lithuanian dairy cattle, to estimate heritability coefficient using multiple traits mixed linear model with permanent environment effects.
2. Research methods
The scientific research was carried out at Lithuanian University of Health Sciences, Veterinary Academy, Department of Animal Breeding, state enterprise Agriculture Information and Rural Business Centre, state enterprise “Pieno tyrimai”, as well as in dairy farms running automatic voluntary milking systems in Lithuania. During the period of 2009-2014 we conducted a research on 421 cows’ data from three dairy farms in Lithuania equipped with automatic voluntary milking systems (Fig. 1). Milking equipment includes robotic manipulator of milking and milk reservoirs. The robot captures the information of each milking: milk yield, milk conductivity, blood in milk, milk flow rate, milking time, teat condition and etc. A total data set of 462574 cow milking records was assessed in the research, 428784 milking’s being from cows’ complete lactations.
Fig. 1Delaval automatic voluntary milking system

The research on the composition of milk (fat, protein, lactose) and the quantity of urea in milk was carried out at the state enterprise “Pieno tyrimai” using milk analyser “LactoScope FTIR” (FT1.0, 2001; Delta Instruments, the Netherlands). This equipment measures the absorption of the specific wavelength of medial infrared rays to determine the amount of each component in milk. Somatic cell count in milk was determined using measuring device “Somascope” (CA-3A4, 2004; Delta Instruments, the Netherlands), which operates on the principle of flow cytometry technology. State enterprise “Pieno tyrimai” operates under quality management system conforming to the requirements of International Standard ISO/IEC 17025:2005 to ensure the accuracy of milk composition and quality tests.
The structure of the data was analysed from three different databases: dairy farms’ herd management programs (Microsoft SQL Server), state enterprises’ Agriculture Information and Rural Business Centre and “Pieno tyrimai” databases (Oracle Database). A new database (Microsoft SQL Server) combining all the research data was created. Descriptive statistic (mean – , standard error –), phenotypic correlation () were performed using the statistical package (The R Project for Statistical Computing, 2012).
PEST [10] software was used to calculate the breeding values of cows by implementing BLUP (best linear unbiased prediction) method model. The research was based on Black-and-White cattle data. Seven indicators (electrical conductivity of milk, milk yield, milk fat, milk protein, lactose, urea, somatic cell count) were analysed using one multivariate test model:
wherw: – observation vector of phenotypic values; – lactation (4 classes, fixed effect); – month of lactation (12 classes, fixed effect); – time of year (2 classes, fixed effect); – milk farm (3 classes, fixed effect); – animal ID (3341 classes, additive-genetic random effect); – repeatability effect (268 classes).
VCE [11] software was used to estimate the heritability and genetic correlation coefficients () of milk traits by implementing BLUP method model. The matrix notation of the model is as follows:
System of equations with known data (variance components of random effects: genetic effects (), environmental effects (), residuals (), incidence matrix ()):
here – covariance matrix:
where – vector of observations; , , – incidence matrix for all fixed, animal and environmental effects, respectively , , – vector of unknown parameters of fixed, animal and environmental effects, respectively; – pedigree matrix; – vector of residuals.
3. Results and discussion
3.1. Descriptive statistic, phenotypic and genetic relationship between traits
The average electrical conductivity of cows’ milk corresponded to healthy cows’ rate, 94 % of records ranged from 3.8 to 6.2 mS/cm (Table 1). In accordance with V. Špakauskas et al. [12], the electric conductivity of milk from cows with subclinical mastitis may increase from 6.1 to 8.5 mS/cm. E. Norberg with a group of scientists [13] found that the average electrical conductivity of healthy cows tested in Denmark was 4.87 mS/cm, when identified with subclinical mastitis – 5.37 mS/cm, clinical mastitis – 6.44 mS/cm. Some researchers [14, 15] argue that electrical conductivity of milk of healthy cows is 4.0-5.5 mS/cm. V. Juozaitienė with scientists [16] found that electrical conductivity of Lithuanian Black and White cows was on average 6.30±0.09 mS/cm and was associated with an indirect indicator of mastitis in cows – an increased number of somatic cells in milk. The majority of somatic cells are leukocytes (white blood cells) – which become present in increasing numbers in milk usually as an immune response to a mastitis-causing pathogen – and a small number of epithelial cells, which are milk-producing cells shed from inside of the udder when an infection occurs. Somatic cell count (SCC) of a cow that is not infected with mastitis is usually less than 200,000 cells/ml and many cows maintain SCC values of less than 100,000 cells/ml. When infection occurs, the macrophages present in the udder signal the cow’s immune system to send neutrophils to engulf and destroy the bacteria. More than 90 % of SCC in infected glands are composed of neutrophils and SCC of greater than 200,000 cells/ml is a strong indicator of mastitis.
The best productivity and milk quality of cows according to somatic cell count was observed in the second milk electrical conductivity class – 4.5-5.5 mS/cm. Changes in indicators are grossed up to 2 and 3 polynomial degree (Figs. 2 and 3).
Table 1Descriptive statistic
Milk trait | M±SE |
Electrical conductivity, mS/cm | 4.92±0.0001 |
Milk yield, kg | 25.12±0.020 |
Fat % | 4.07±0.0001 |
Protein % | 3.47±0.0001 |
Lactose % | 4.63±0.0001 |
Urea mg % | 23.76±0.013 |
Somatic cell count 103/ml | 188.95±1.032 |
Fig. 2Dependence of productivity of cows on electrical conductivity of milk
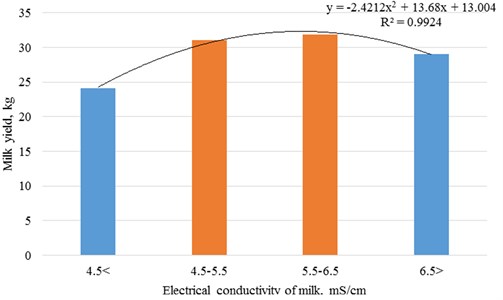
Fig. 3Dependence of somatic cell count on cows’ electrical conductivity of milk
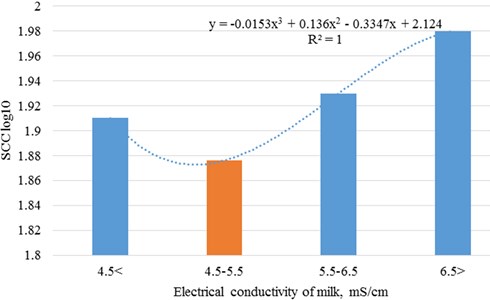
The research of electrical conductivity of milk in Lithuanian dairy cattle herds has shown that electrical conductivity of milk ranged from 4.6 to 5.8 mS/cm in milk samples where somatic cell count did not exceed 200 thousand/ml.
The largest amount of milk protein fat (; ) and lactose (; ) were found in milk samples of class 1 and linear decrease of traits by class of milk electrical conductivity was observed (Fig. 4).
The control of variability in milk urea concentration (MUC) is used for a diagnosis of the energy-nitrogen metabolism of cows [17, 18]. Both the high and the low levels of MUC may exert a negative effect on the health of dairy cows [19]. According to the data of investigations (Table 1) the average of milk urea (23.76±0.013) was within the normal (15-30 mg %) range [20].
The phenotypic and genetic correlations between electrical conductivity, milk yield, milk composition and SCC are summarised in Fig. 5.
Fig. 4Dependence of milk fat, protein and lactose amount on electrical conductivity of milk
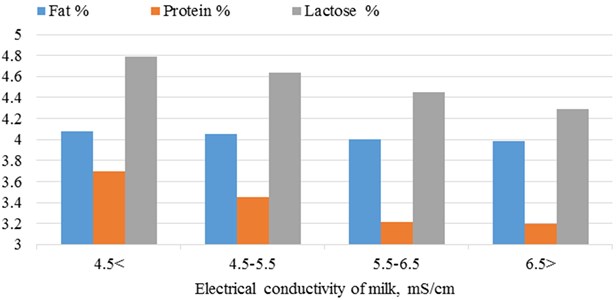
Fig. 5Phenotypic and genetic relationship of electrical conductivity with other milk traits
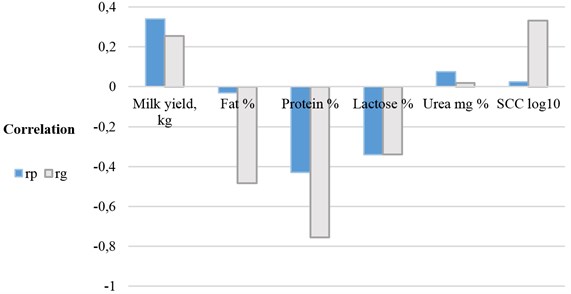
After evaluation of phenotypic dependence between traits we found positive correlation of the electrical conductivity of milk with milk production (0.341), negative correlation with milk protein (–0.431) and lactose (–0.340) (0.001). Electrical conductivity of milk has expressed a positive genetic correlation with milk yield (0.256) and somatic cell count (0.332), a negative genetic correlation with milk fat (–0.485), milk protein (–0.757) and lactose ( –0.34), ( 0.001).
If a cow has mastitis, the concentration of Na- and Cl- in the milk increases due to damage of cells in the mammary tissue. This leads to increased EC of milk from the infected quarters [21]. A large amount of research has been conducted to validate EC as an indicator trait for mastitis, and a clear phenotypic association between the traits has been found [13, 22]. E. Norberg et al. [23, 24] found a genetic correlation between the level of EC and clinical mastitis to be 0.75, between EC and SCC – 0.06-0.89.
3.2. Heritability of milk traits
We have found a low (0.032) heritability of SCC (Fig. 6). This genetic parameter for SCC estimated in this research is in agreement with results (0.07-0.12) from other studies [8, 9]. The heritability for SCC is low and little genetically progress can be made toward decreasing the somatic cell count in milk. The low heritability of SCC will result in a slow response to selection for resistance to mastitis.
A high coefficient of heritability of electrical conductivity of milk ( 0.512 ± 0.028; 0.001) was determined by multivariate model in our research. This coefficient was the highest of all investigated indicators. A similar result of EC ( 0.56) was received by A. Gáspárdy with a group of researchers [25]. In the United States using an animal model that included herd-test day, age at calving and days in milk as fixed effects, and random additive genetic and permanent environmental effects data of EC from 2101 first-lactation Holstein cows in 8 herds was analysed. E. Norberg et al. [23] estimated the heritability of EC to be in the range 0.26 to 0.36 in this model. Due to the relatively high heritability obtained for EC of milk, EC might be a potential indicator trait to use in a breeding program designed to reduce the incidence of mastitis. Heritability for EC in Italian Brown cattle [7] was equal to 0.23 (115,799 milking records involving 429 lactations and 233 cows were available).
Fig. 6Heritability of milk traits
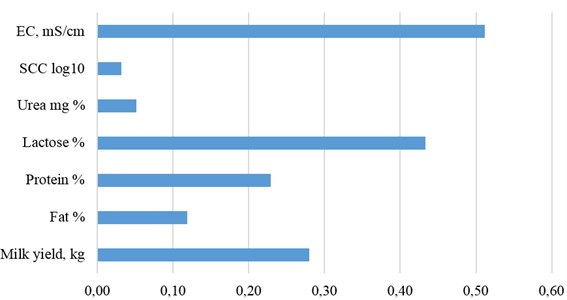
These results show that genetic improvement can be achieved in SCC by including EC in the selection program. Therefore, the EC trait can be a beneficial indicator in detecting mastitis and should be considered in sire selection. In addition to a high heritability, EC may have some advantages compared to somatic cell count (SCC) in terms of cost and frequency of registrations. Electrical conductivity is easily measured during milking, and repeated records from each day during the lactation may be utilised for genetic evaluation.
The robotic milking system is a new style of farm management whereby the decisions are transferred from the farmer to the cow. This system software assists in heat detection, rumination, SCC levels, milk weights and individual grain feeding. These abilities need consideration for cost-benefit analysis. They cannot discard milk from individual quarters. We noticed that robotic milking system influenced on milk conductivity and this parameter was as indicator of SCC level in milk. Equal or improved somatic cell counts, herd health and reproduction with increased management ability.
Changes in electrical conductivity of milk can be used for the purpose of monitoring mastitis status of dairy cows [6, 13, 26-28].
Electrical conductivity of milk from robotic milking system can been introduced as an indicator of mastitis prevention in dairy cows and genetic selection based on this trait may be possible.
4. Conclusions
Application of robotic milking system can have a positive effect on milk conductivity and conductivity may affect the possibility for early diagnosis of mastitis, without prejudice to the cow's health.
The ability to record data of electric conductivity of cows’ milk on-line, high heritability coefficient of electrical conductivity of milk and electrical conductivity of milk can be used in dairy cattle breeding programs aimed at effectively improving productivity and cows’ health. Positive genetic correlation between electrical conductivity of milk and somatic cell count, as determined in the research, indicate that in order to diagnose cows with mastitis in its early stage, it is appropriate to monitor the changes of electrical conductivity of cows’ milk.
References
-
Vorobjovas G., Žilaitis V., Banys A., et al. Automated cows milking influence on quantity of milk and its composition. Veterinary Medicine and Zootechnics, Vol. 51, Issue 73, 2010, p. 71-76, (in Lithuanian).
-
Svennersten-Sjaunja K. M., Pettersson G. Pros and cons of automatic milking. Journal of Animal Science, Vol. 86, 2008, p. 37-46.
-
Bloemhof S., de Jong G., de Haas Y. Genetic parameters for clinical mastitis in the first three lactations of Dutch Holstein cattle. Veterinary Microbiology, Vol. 134, 2009, p. 165-171.
-
Norberg E. Electrical conductivity of milk as a phenotypic and genetic indicator of bovine mastitis: a review. Livestock Production Science, Vol. 96, 2005, p. 129-139.
-
Hovinen M., Aisla A. M., Pyörälä S. Accuracy and reliability of mastitis detection with electrical conductivity and milk colour measurement in automatic milking. Acta Agriculturae Scand Section A, Vol. 56, Issue 3-4, 2006, p. 121-127.
-
Ilie L. I., Tudor L., Galis A. M. The electrical conductivity of cattle milk and the possibility of mastitic diagnosis in Romania. Journal of Veterinary Medical Science, Vol. 43, 2010, p. 220-227.
-
Povinelli M., Gallo L., Carnier P., et al. Genetic aspects of milk electrical conductivity in Italian Brown cattle. Italian Journal of Animal Science, Vol. 4, Issue 3, 2005, p. 169-171.
-
Mrode R. A., Swanson G. J. T. Estimation of genetic parameters for somatic cell counts in the first three lactations using random regression. Livestock Production Science, Vol. 79, Issue 2, 2003, p. 239-247.
-
Ødegård J., Jensen J., Klemetsdal G., et al. Genetic analysis of somatic cell score in Norwegian cattle using random regression test-day models. Journal of Dairy Science, Vol. 86, Issue 12, 2003, p. 4103-4114.
-
Groeneveld E., Kovac M., Wang T. PEST Users Manual Guide. Institute of Animal Husbandry and Animal Behavior, FAL, Mariensee, Germany, 1998.
-
Groeneveld E. VCE 4 Version 4.2.5. Institute of Animal Husbandry and Animal Behavior, FAL, Mariensee, Germany, 1998.
-
Špakauskas V., Klimienė I., Matusevičius A. A comparison of indirect methods for diagnosis of subclinical mastitis in lactating dairy cows. Veterinarski Archive, Vol. 76, Issue 2, 2006, p. 101-109.
-
Norberg E., Hogeveen H., Korsgaard I. R., et al. Electrical conductivity of milk: ability to predict mastitis status. Journal of Animal Science, Vol. 87, 2004, p. 1099-1107.
-
Hillerton J. E., Walton A. W. Identification of subclinical mastitis with a hand-held electrical conductivity meter. Veterinary Record, Vol. 128, Issue 22, 1991, p. 513-515.
-
Hamann J., Gyodi P. Somatic cells and electrical conductivity in relation to milking frequency. Milchwissensch, Vol. 55. 2000, p. 303-307.
-
Juozaitienė V., Šlapkauskaitė J., Tušas S., et al. Electrical conductivity changes of milk during milking phase with productivity and somatic cell count. Veterinary Medicine and Zootechnics, Vol. 51, Issue 73, 2010, p. 23-29.
-
Jilek F., Řehak D., Volek J., et al. Effect of herd, parity, stage of lactation and milk yield on urea concentration in milk. Czech Journal of Animal Science, Vol. 51, 2006, p. 510-517.
-
Zhai S. W., Liu J. X., Wu Y. M., et al. Responses of milk urea nitrogen content to dietary crude protein level and degradability in lactating Holstein dairy cows. Czech Journal of Animal Science, Vol. 51, 2006, p. 518-522.
-
Kubešova M. Frelich J. Maršalek M. Relation of milk components content and reproduction indexes in dairy cows. Collection of Scientific Papers Faculty of Agriculture in Česke Budějovice Series for Animal Sciences, Vol. 2, 2005, p. 229-238.
-
Sederevicius A., Kabasinskiene A., Savickis S., et al. Milk urea nitrogen as an important indicator of dairy cow nutrition. Review. Veterinary Medicine and Zootechnics, Vol. 44, Issue 66, 2008, p. 23-30.
-
Kitchen B. J. Review of the progress of dairy science: Bovine mastitis. Milk compositional changes and related diagnostics tests. Journal of Dairy Research, Vol. 48, 1981, p. 167-172.
-
Hamann J., Zecconi A. Evaluation of the Electrical Conductivity of Milk as a Mastitis Indicator. Bulletin of the International Dairy Federation, Vol. 334, Brussels, Belgium, 1998.
-
Norberg E., Rogers G. W., Goodling R. C., et al. Genetic parameters for test-day electrical conductivity of milk for first-lactation cows from random regression models. Journal of Animal Science, Vol. 87, Issue 6, 2004, p. 1917-1924.
-
Norberg E., Ødegård J., Madsen P. Comparison of variance components for test-day electrical conductivity of milk and test-day somatic cell score for first lactation cows in an experimental herd. Acta Agriculturae Scandinavica, Section A-Animal Science, Vol. 54, Issue 4, 2004, p. 181-186.
-
Gáspárdy A., Ismach G., Bajcsy Á. C., et al. Evaluation of the on-line electrical conductivity of milk in mastitic dairy cows. Acta Veterinaria Hungarica, Vol. 60, Issue 1, 2012, p. 145-155.
-
Kamphuis C., Pietersma D., Van der Tol R., et al. Using sensor data patterns from an automatic milking system to develop predictive variables for classifying clinical mastitis and abnormal milk. Computers and Electronics in Agriculture, Vol. 62, Issue 2, 2008, p. 169-181.
-
Mottram T., Rudnitskaya A., Legin A., et al. Evaluation of a novel chemical sensor system to detect clinical mastitic in bovine milk. Biosensors and Bioelectronics, Vol. 22, 2007, p. 2689-2693.
-
Kasikci G., Cetin O., Bingol E. B., et al. Relations between electrical conductivity, somatic cell count, California mastitis test and some quality parameters in the diagnosis of subclinical mastitis in dairy cows. Turkish Journal of Veterinary and Animal Sciences, Vol. 36, Issue 1, 2012, p. 49-55.
