Abstract
The results in vivo investigation biophotonics of laser-induced photodissociation of oxyhemoglobin in cutaneous blood vessels and its role in biomedical processes are presented. It is shown that in order to make the methods of phototherapy as well as laser therapy really efficient one has to control the oxygen concentration in tissue keeping it at the necessary level. Perspectives of combined laser-ultrasound application for improving oxygen diffusion are discussed.
1. Introduction
Biophotonics of "laser-tissue" interaction and the effect of laser radiation on oxyhemoglobin in cutaneous blood vessels and capillaries are considered to be interesting aspects of modern photomedicine and photobiology. Application of low intensity laser radiation in treatment of a variety of diseases was being extensively developed during the last five decades.
Biostimulation and therapeutic effect of laser radiation is a well-established fact, which is currently widely used in clinical practice. At the same time the mechanism of therapeutic effect of laser radiation was not clearly understood for a long time and considered to be very complex and accompanied by anti-inflammatory, analgesic and anti-inflammatory effect on tissue [1-4]. The most exciting effect of laser therapy could be seen in wound healing where a process of fast epithelization clearly demonstrated its efficiency. Since 1998 a new technology of laser-induced photodissociation of oxyhemoglobin (HbO2) in cutaneous blood vessels and its biomedical applications has been developing. The efficiency of the interaction of laser radiation in different wavelengths on HbO2 in cutaneous blood vessels is studied. Mathematical model for calculating optimal parameters of laser radiation to induce an effective photodissociation of hemoglobin (Hb) complexes in cutaneous blood vessels has been developed [5-8].
2. The phenomenon of laser-induced blood oxyhemoglobin hotodissociation
It’s a well-known fact that the concentration of oxygen is critical in enhancing a wide variety of biochemical reactions in vivo, including cell metabolism. Aerobic cell metabolism is a primary mechanism of energy production in tissue. Controlling this mechanism gives a unique possibility of biological stimulation to reach therapeutic effect. This goal could be reached by the means of laser-induced photodissociation of oxyhemoglobin in cutaneous blood vessels.
Absorption of light by blood Hb and HbO2 results in to primarily physical processes: nonradiative dissipation of electronic excitation energy through heat and photodissociation of HbO2 in blood vessels. In case of low laser radiation and estimations show that in a typical case the local increase of temperature of 0.1-0.5 °C may be expected. Such a small rise of a local temperature may promote only some improvement in capillary microcirculation of blood and hardly could stimulate the metabolism of cells.
In case of low energy lasers the most important process is the photodissociation of HbO2, which main biological function is molecular oxygen transport. The quantum efficiency of the photodissociation [9] of oxyhemoglobin is amazingly high and reaches 10 % in a wide visible spectral range. The molecular oxygen is generated due to laser-induced photodissociation of HbO2 in blood vessels, which allows to control the local increase of oxygen concentration at irradiating region (Fig. 1).
Fig. 1Illustration of laser-induced tissue oxygenation caused by photodissociation of arterial blood HbO2
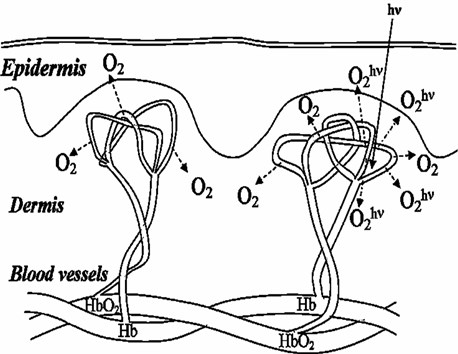
The possibility of additional oxygen supply allows to develop a new method of tissue hypoxia elimination that restores normal cell metabolism. Investigation of photodissociation of hemoglobin complexes in vivo could be carried out using arterial blood saturation parameter.
For HbO2 the value of saturation SaO2 in arterial blood vessels is defined by the concentration of HbO2 taking into account contribution of Hb, methemoglobin (MetHb) and carboxyhemoglobin (HbCO).
At normal conditions of gas exchange the concentrations of MetHb and HbCO are extremely low (0.2-0.6 % and 0.8 % correspondingly) so the contribution of these components can be neglected. Thus in practice the value of SaO2 could be determined as:
Photodissociation of HbO2 induced by laser radiation releases free molecular oxygen. Meanwhile, the proportion between [HbO2] and [Hb] concentrations is changed which decreases the value of SaO2.
where SaO2 is saturation without and SaO2hwith laser irradiation.
Amount of oxygen available for cell metabolism delivered by microcirculation is the function of:
where HbO2 is the value of oxyhemoglobin arterial blood and [O2] – is the concentration of oxygen released into plasma.
In case of deterioration of the blood microcirculation extra oxygen supply is critical to provide the demands of cell for normal metabolism. This could be reached by in vivo laser-induced photodissociation of HbO2 directly to the zone where it is necessary to increase local concentration of free molecular oxygen.
As a result we obtain average concentration of oxygen releasing in a conventional way and due to:
Thus the phenomenon of laser-induced in vivo photodissociation of oxyhemoglobin in cutaneous blood vessels and capillaries gives a unique possibility of optical increase in local tissue oxygen concentration.
3. Experimental investigation
The method of transcutaneous oximeter that is based on the principle of measuring oxygen tension РО2 in arterial blood is a direct method of registration of gases that dissolved in blood plasma.
For this purpose one usually uses Clark-type polarographic sensor (TcPO2 electrode) that consist of a silver anode, electrolyte, and an oxygen permeable membrane; heating section and electronic system for measuring and controlling the sensor temperature.
The initial oxygen tension in tissue was measured by placing TcPO2 electrode on human skin in shoulder area. Then He-Ne laser radiation at the power of 1mW was applied. Kinetics of tissue oxygen tension was experimentally investigated [10]. The results obtained were normalized to the initial oxygen tension value.
These methods mentioned above allow to measure and control the process of releasing extra oxygen from HbO2 under laser irradiation directly to arterial blood plasma and further it diffusion into tissue.
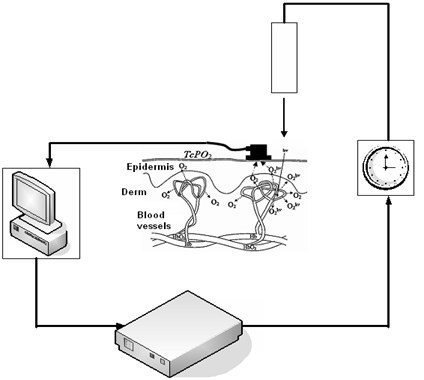
Experimental investigation of laser-induced tissue oxygenation phenomenon has been carried out using transcutaneous oxygen monitor (TCOM) – "Radiometer” ТСМ 4 (Fig. 2). Direct in vivo measurements of tissue oxygen tension TcPO2 under irradiation by He-Ne laser at the power of 1 mW has been taken [10].
Fig. 2Experimental setup for controlling local tissue oxygen concentration directly at the zone of laser irradiation
Using a simple diffusion model we calculated what amount of oxygen should be released into blood plasma in order to reach experimentally observed tissue O2 concentration (Fig. 3). The target criteria were kinetics of tissue oxygenation in response to laser irradiation.
The variable parameters were diffusion coefficient of oxygen in tissue and oxygen release rate.
Fig. 3Simple model of oxygen diffusion in tissue
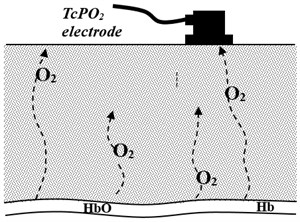
As it was shown experimentally [8] the response of oxygen release on laser irradiation is relatively fast and remains constant during the irradiation. To simulate this effect in the model, the oxygen release rate was increased instantly and remained constant during the time of irradiation.
The main aim of the calculation was to reach the closest fit of the data produced by the model to the experimentally measured one. The target criteria were kinetics of tissue oxygenation in response to laser irradiation. The variable parameters were diffusion coefficient of oxygen in tissue and oxygen release rate.
4. Results and discussion
The kinetics of oxygen tension in tissue in two cases including normal blood circulation and artificially induced ischemia was investigated. The obtained results were normalized to initial oxygen tension value.
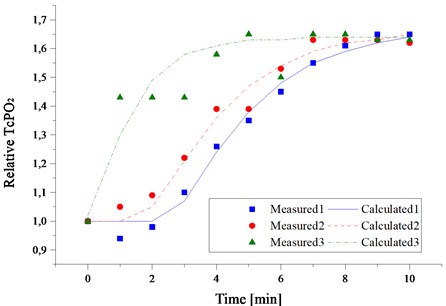
In Fig. 4 the experimental results of in vivo laser induced tissue oxygenation for volunteer in normal condition and in case of artificially induced ischemia are presented. As it can be seen we still can extract extra oxygen from arterial blood and optically satisfy the cell metabolism demand for the required period of time.
As it can be seen from Fig. 4 after the 10 minutes of exposure under laser irradiation, the value TcPO2 reaches saturation level which is approximately 1.6 times higher than initial. In all three cases, the calculations demonstrate practically the same increase (about 4.3 times) of the value of oxygen release rate under the irradiation by laser light but calculated diffusion coefficient of oxygen in tissue differs significantly (up to two times). This demonstrates individual differences in density of skin.
Fig. 4The kinetics of laser-induced tissue oxygenation during laser irradiation measured on three different individuals
Supplemental oxygen can lead to the increased rate of collagen deposition, epithelization and improved healing of split thickness grafts. Increased subcutaneous TcPO2 has also been shown to improve bacterial defences. It should be noted that often biological tissue affected by disease characterised by damaged or disorganised the blood capillary network. Thus diffusion plays an important role in oxygen delivery in such tissue.
So an important conclusion can be drawn from the obtained results. Increasing diffusion rate in affected area will significantly improve efficiency of the phototherapy. A promising approach to increase diffusion rate in the tissue is to apply ultrasound waves to the affected area. According [11] ultrasound exposure of 1…2 Wcm-2 increases significantly the rate of oxygen transport through the skin.
Combined Laser and Ultrasound action that enriches tissue oxygenation has a potential to become novel therapeutical method for treatment of the pathologies where elimination of local tissue hypoxia is critical.
5. Conclusion
The new optical method of local tissue hypoxia elimination has been demonstrated. The value of tissue oxygen concentration increases significantly during the laser irradiation.
The method of determination of oxygen diffusion coefficient on kinetics of tissue O2 under the laser irradiation has been developed.
It has been shown that the efficiency of laser-induced oxygenation is comparable to the method of hyperbaric oxygenation gaining advantages at the same time in local action.
New method of increasing efficiency of the phototherapy by of combining laser irradiation with ultrasound tissue activation has been proposed.
Photodissociation of oxyhemoglobin, the main biological function of which is molecular oxygen transport gives a unique possibility of additional oxygen supply and allows develop laser-optical method of tissue hypoxia elimination that restores normal cell metabolism.
In order to make the phototherapy as well as laser therapy methods really efficient one has to control the O2 concentration in tissue keeping it at the necessary level. This goal could be reached by the use of laser-induced photodissociation of HbO2 in tissue blood vessels.
References
-
Takas S., Stojanovich S. Diagnostic and biostimulating lasers. Med. Pregl., Vol. 51, Issue 5-6, 1998, p. 245-249.
-
Baxter G. D. Therapeutic lasers: Theory and Practice. Edinburgh, New-York, 1994.
-
Tuner J., Hode L. Laser Therapy: Clinical practice and scientific background. Prima Books AB, 2002.
-
Mostovnikov V. A., Mostovnikova G. R., Plavski V. Yu, Plavskaya L. G. Biophysical principles of regulatory action of low-intensity laser irradiation. Proc. SPIE., Vol. 2728, 1996, p. 50-62.
-
Asimov M. M., Asimov R. M., Rubinov A. N. Investigation of the efficiency of laser action on hemoglobin and oxyhemoglobin in the skin blood vessels. Proc. SPIE., Vol. 3254, 1998, p. 407-412.
-
Asimov M. M., Asimov R. M., Rubinov A. N. Action spectra of laser radiation on hemoglobin of skin blood vessels. Journal of Applied Spectroscopy, Vol. 65, Issue 6, 1998. p. 877-880.
-
Asimov M., Asimov R., Mirshahi M., Gizbrebrecht A. Effect of laser induced photodissociation of oxyhemoglobin on biomedical processes. 11th International School on Quantum Electronics: Laser physics and Applications, Proc. SPIE., Vol. 4397, 2001, p. 390-394.
-
Asimov M. M. Laser-induced Photodissociation of Hemoglobin Complexes with Gas Ligands and its Biomedical Applications. Proc. of LTL Plovdive 2005, IV International Symposium Laser Technologies and Lasers, Plovdive, Bulgaria, 2005, p. 3-11.
-
Saffran Wilma A., Quentine H. Gibson Photodissociation of Ligands from Hem and Hem Proteins: Effect of Temperature and Organic Phosphate. Journal of Biol. Chem., Vol. 252, 1977, p. 7955-7958.
-
Asimov M. M., Korolevich A. N., Konstantinova E. E. Investigation of the kinetics of tissue oxygenation under the effect of low intensity laser radiation. Journal of Appl. Spectr., Vol. 74, 2007, p. 120-125.
-
Mortimer A. J., Trollope B. J., Villeneuve E. J., Roy O. Z. Ultrasound-enhanced diffusion through isolated frog skin. Ultrasonics, Vol. 26, Issue 6, 1988, p. 348-351.
