Abstract
An alteration of the cognitive functions plays a crucial role in the quality of life in the elderly. New investigations notice that the thickness of carotid intima-media (CIMT) might help to identify patients with an increased risk of cognitive deterioration. Our aim of the study was to evaluate CIMT of the patients with mild cognitive impairment (MCI) with static (manually) and dynamic (RF-QIMT) ultrasonographic equipments. Our results show that the CIMT was thicker measuring by semi-automated method, than manually and the differences were statistically significant. Our results show no correlation between the CIMT and scores of psychiatric tests, therefore more research is needed for improvement of carotid wall thickening changes in MCI patients.
1. Introduction
The measurement of the carotid intima-media thickness (CIMT) is recognized as potential vascular marker of atherosclerosis and is an independent predictor of future vascular events [1, 2, 21]. Additionally, new studies indicate, that an increased CIMT predicts a cognitive decline in an elderly population [14, 15, 22, 23]. If the patient has memory complaints, objective memory impairment for age, preserved general cognition, normal activities of daily living and is not demented, mild cognitive impairment (MCI) can be diagnosed [3, 4]. Unfortunately, about 10-15 % of MCI converse annually to a dementia [3-5].
There are three types of CIMT measurement: (1) conventional B-mode scanning, (2) semi-automated and fully automated [28, 34]. However, the evaluation of CIMT in many of them in frozen or static ultrasound image is performed. To this date only Radio Frequency-Quality Intima Media Thickness (RF-QIMT) is worldwide accepted as safe and trusted dynamic evaluation of CIMT [32, 33]. Despite increasing literature data of MCI and CIMT correlation, there is a lack of studies using the RF-QIMT method [20, 32, 33]. Additionally, a limited number of studies have examined the association between static and dynamic CIMT measurements [32]. Therefore our aim of the study was to evaluate CIMT of MCI patients with static (manually) and dynamic (RF-QIMT) ultrasonographic equipments.
2. Materials and methods
The study was performed at the Lithuanian University of Health Sciences Clinical Hospital in the period of 2011-2012 years. In the study were included those patients who fit MCI diagnostic criteria [3, 4]. Patients with chronic cardiac insufficiency, signs of dementia or brain ischemic injury were not included. All patients underwent computed tomography or magnetic resonance imaging of the brain to exclude tumor, ischemic or postoperative changes.
The Mini Mental State Examination (MMSE) and Alzheimer’s Disease Assessment Scale-Cognitive subscale (ADAS-Cog) tests were employed as a measure of global cognitive function. All the patients underwent a structural interview, neurological examination and laboratory tests to exclude secondary causes of cognitive impairment. Additionally, body mass index (BMI) was assessed. The written consent was obtained from each patient in order to be involved into the study. The local Ethics Committee approved this latest one.
Fig. 1Carotid intima-media thickness measurement with ImageJ software (a) and ArtLab system (b)
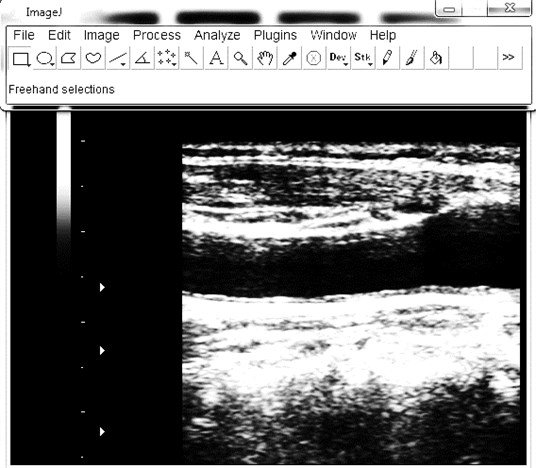
a)
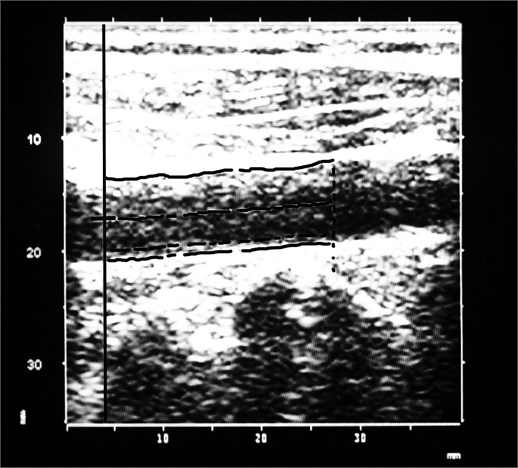
b)
The scanning of carotid arteries with ultrasound equipment (Voluson 730 KretzTechnic, GE Medical Systems, Austria), including a high-resolution B-mode system with linear ultrasound 7 MHz transducer, was performed. The depth focus, frame rate and gain settings in standard manner were used to each patient. The measurement of CIMT 1 cm below the carotid bulb in a region free of plaque, according to Mannheim consensus and other guidelines was performed [6, 7]. The thickness of intima-media was measured on the far wall in six places along a minimum 10 mm of an arterial segment (Fig. 1a). The ultrasound images of the left and right carotid arteries were archived and for more accurate results were analyzed with freely available ImageJ software (National Institutes of Health, USA, http://imagej.nih.gov/ij). The same segment of the far wall of carotid artery was scanned with Picus Pro (Esaote, Netherlands) ultrasound device with an automatic boundary detection system (ArtLab, Netherlands) (Fig. 1b). ArtLab system automatically calculated the CIMT from six places. For excluding bias, one researcher with five years of experience made all measurements. For statistical analysis, only mean values of one measurement were used. Atherosclerotic changes of the carotid wall were not included in the calculation.
Statistical calculations were performed with MedCalc software (MedCalc, Mariakerke, Belgium, version 11.4.2.0) using Bland-Altman plot, Pearson's correlation coefficient. D’Agostino-Pearson test for normal distribution was used. Mann-Whitney test was used when the sample data was not normal. Student’s t criterion was used when the data was normally distributed. All the values were given as a mean and standard deviation. 0.05 was considered statistically significant (confidence interval (CI) – 95 %).
3. Results
All examinations were performed on 90 patients. However, only 77 patients were included in the study, because nine residual patients had additional psychiatric disorders, like severe dementia, organic mental disorders and other. Four patients had the class II or III heart failure according to the New York Heart Association (NYHA) classification.
The mean age was 67.9±9.1 years, 18 (23 %) were males and 59 (77 %) females. BMI was 29.1±5.2 kg/m2. The mean of MMSE score was 25.3±2.5 points (range 18-30), respectively the mean of ADAS-Cog score was 17.0±6.0 (range 5-30).
We measured in total 143 common carotid arteries by ArtLab (mean 793±118 µm, range 532-1134 µm) and 154 by ImageJ (mean 766±157 µm, range 452-1160 µm).
The data points of the measurements by ArtLab and ImageJ were relatively close, but not ideally on a regression line, which suggested some degree of disagreement between the two methods, however the Pearson’s correlation coefficient between CIMT was strong ( 0.64, 0.0001; 0.53, 0.73, 143) (Fig. 2a). The difference between the ArtLab and ImageJ measurements was -26,9 µm, with a 95 % confidence interval from -58,5 to 4,7 µm and an associated P value of 0.0945. Median latencies in ArtLab and ImageJ were 787 and 745 µm; the distributions in the two measurement groups differed significantly (Mann–Whitney 9541.5, Test statistic 1.99, 0.0469 two-tailed). The data comparison graphs of ArtLab and ImageJ measurements are represented in Figure 2b.
We established strong correlations comparing ArtLab and ImageJ of right side CIMT ( 0.68, 0.0001; 72); right ArtLab and left ArtLab ( 0.66, 0.0001; 70); right ArtLab and left ImageJ ( 0.50, 0.0001; 72), right ImageJ and left ArtLab ( 0.41, 0.0004; 71), right ImageJ and left ImageJ ( 0.70, 0.0001; 77), left ArtLab and left ImageJ ( 0.60, 0.0001; 71).
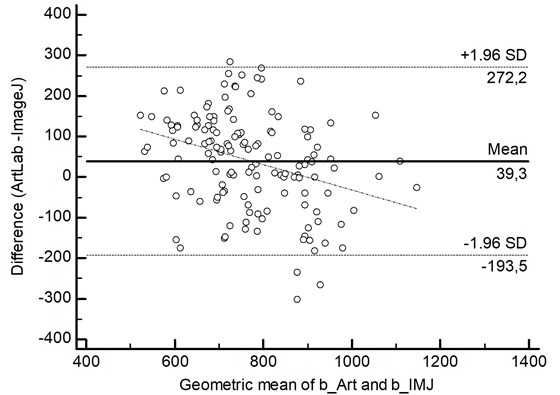
In 2.8 % (4 of 143 cases) of the data points were outliners and exceeded the upper limit of agreement (Fig. 3).
The bias of the CIMT measurements was 39.3 ( 19.7, 60.0), indicating that by ArtLab method slightly higher values of the CIMT than by ImageJ method were measured. The precision was 118.8 (95 % confidence limit -193.5, 272.2 µm).
Fig. 2The measurements of carotid intima-media thickness with ArtLab system and ImageJ software are representing: a) scatter diagram, b) data comparison graph
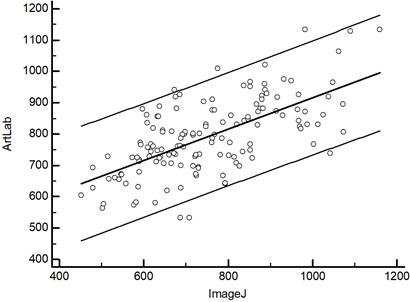
a)
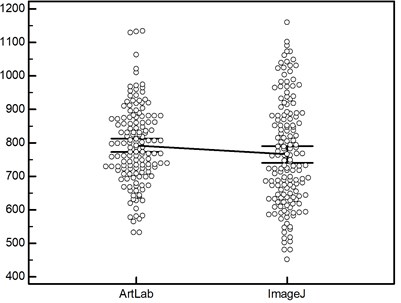
b)
Fig. 3Bland-Altman plot of carotid intima-media thickness differences, measured by ArtLab system and ImageJ software. The dotted line represents systematic error
We found medium correlations comparing age and right ArtLab ( 0.33, 0.0038; 72), left ArtLab ( 0.26, 0.0321; 71). All carotid ArtLab ( 0.30, 0.0003; 143), right ImageJ ( 0.41, 0.0002; 77), left ImageJ ( 0.34, 0.0026; 77), all carotid ImageJ ( 0.38, 0.0001; 154), ADAS-Cog ( 0.44, 0.0001; 77), MMSE ( -0.30, 0.0087; 77). Additionally, we established negative strong correlations between ADAS-Cog and MMSE ( -0.56, 0.0001; 77). We did not establish other significant correlations, including BMI, psychiatric diagnostic tests and CIMT.
4. Discussion
An alteration of the cognitive functions plays a crucial role in the quality of life in the elderly. The studies have shown the influence of the brain small vascular diseases, high blood pressure, diabetes mellitus, hypercholesterolemia, atherosclerosis to cognitive impairment [8]. Additionally, new investigations notice that the thickness of carotid intima-media (CIMT) might help to identify patients with an increased risk of cognitive deterioration [11].
The first choice of imaging technique of extra cranial arteries is high-resolution B-mode and pulsed Doppler ultrasonography. With this technique we can easily asses the CIMT, the morphology of atherosclerotic plaques, location / size of the stenosis and other. For lowering the influence of researcher, the manual measurement of CIMT can be changed by semi-automated and fully automated software. However, semi-automated systems can not perform without human interventions, therefore a small influence still remains. New promising fully automated programmes are already underway, however more evidence and time is needed to improve them into clinical practice.
In our study we used manual and semi-automated techniques for measuring CIMT. Although the manual measurement is long lasting, with large interpretation errors and depending from researchers experience, however many studies about the relation of CIMT and dementia used this method [22]. Additionally we use a semi-automated RF-QIMT technique, based on classic high resolution echo-tracking technology including the use of a 128 radio frequency line multiarray [20]. In literature there is lack of information according to the use of this new technique. Arntzen K. A. and co-authors have analyzed the CIMT, however they used another type of semi-automated method. Moreover, we did not find any information about the RF-QIMT measurement of CIMT of MCI patients.
Our results show, that the CIMT was thicker when measured by semi-automated (ArtLab) method, than manually (ImageJ) and the difference was statistically significant. Shreuder F. H. et al. [32] found different results. They pointed out that the CIMT measured in 136 patients by RF-QIMT was significantly smaller. They explain the difference between both methods due to advanced atherosclerosis. However our correlations and the CIMT of both methods are quite similar: authors Pearson 0.765, B-mode – 779±196 µm, RF-QIMT – 734±172 µm; our study Pearson 0.64, B-mode – 766±157 µm, RF-QIMT – 793±118 µm. We believe that more researches are needed to clarify our difference of results and we agree with opinion, that both methods are useful to measure CIMT in clinical practice. Another study confirms the high accuracy of RF-QIMT in evaluation of patient with coronary artery disease [33].
In our study we did not find any CIMT correlations with the cognitive test scores. However, literature data show, that an increased CIMT predicts a cognitive decline after some follow-up period [22, 23]. We cannot compare our results with these cohort studies, because we used cross-sectional study design. Therefore more research is needed to improve CIMT role in MCI development.
A recent review by Arntzen K. A. et al. [24] pointed out the representation of CIMT as a hypertensive hypertrophic response of smooth muscle cells to changes in local shear and tensile stresses to the vessel wall. Thus atherosclerotic plaques are mostly found at the bifurcation site or in the proximal part of internal carotid artery, where non-laminar turbulent flow occurs. In our study we made all measurements only in distal common carotid artery part without plagues. We did not analyze atherosclerotic plaque incidence. However our results suggest that we cannot detect an initial changing of carotid arteries walls of MCI patients neither with B-mode, nor with RF-pulse ultrasonography imaging.
In conclusion we would like to point out that the measurement of carotid wall with radiofrequency ultrasonography is time saving and more accurate imaging modality than measuring manually with ImageJ software. Our results show no correlation between the CIMT and scores of psychiatric tests, therefore more research needs to be done for improvement of carotid wall thickening changes in MCI patients.
References
-
Gepner A. D., Wyman R. A., Korcarz C. E. et al. An abbreviated carotid intima-media thickness scanning protocol to facilitate clinical screening for atherosclerosis. J. Am. Soc. Echocardiogr., Vol. 20, 2007, p. 1269-1275.
-
Skilton M. R., Serusclat A., Harrisch A.et al. Noninvasive measurement of carotid extra-media thickness. JACC: Cardiovascular Imaging, Vol. 2, 2009, p. 2176-2182.
-
Petersen R. C., Negash S. Mild cognitive impairment: an overview. CNS Spectr., Vol. 13, Issue 1, 2008, p. 45-53.
-
Petersen R. C. Mild cognitive impairment as a diagnostic entity. J. Intern. Med., Vol. 256, 2004, p. 183-194.
-
Grundman M., Petersen R. C., Ferris S. H. et al. Alzheimer's disease cooperative study. Mild cognitive impairment can be distinguished from Alzheimer disease and normal aging for clinical trials. Arch. Neurol., Vol. 61, Issue 1, 2004, p. 59-66.
-
Touboul P. J., Hennerici M. G., Meairs S. et al. Mannheim carotid intima-media thickness consensus (2004-2006). An update on behalf of the Advisory Board of the 3rd and 4th Watching the Risk Symposium, 13th and 15th European Stroke Conferences, Mannheim, Germany, 2004, and Brussels, Belgium, 2006. Cerebrovasc. Dis., Vol. 23, Isseu 1, 2007, p. 75-80.
-
Wikstrand J. Methodological considerations of ultrasound measurement of carotid artery intima-media thickness and lumen diameter. Clin. Physiol. Funct. Imaging, Vol. 27, Issue 6, 2007, p. 341-345.
-
Hurst R. T., Ng D. W., Kendall C., Khandheria B. Clinical use of carotid intima-media thickness: review of the literature. J. Am. Soc. Echocardiogr., Vol. 20, Issue 7, 2007, p. 907-914.
-
Viticchi G., Falsetti L., Vernieri F. et al. Vascular predictors of cognitive decline in patients with mild cognitive impairment. Neurobiol. Aging, Vol. 33, Issue 6, 2012, p. 1127.
-
Cardenas V. A., Reed B., Chao L. L. et al. Associations among vascular risk factors, carotid atherosclerosis, and cortical volume and thickness in older adults. Stroke, Vol. 43, Issue 11, 2012, p. 2865-2870.
-
Gorelick P. B., Scuteri A., Black S. E. et al. American Heart Association Stroke Council, Council on Epidemiology and Prevention, Council on Cardiovascular Nursing, Council on Cardiovascular Radiology and Intervention, and Council on Cardiovascular Surgery and Anesthesia. Vascular contributions to cognitive impairment and dementia: a statement for healthcare professionals from the American Heart Association/American Stroke Association. Stroke, Vol. 42, Issue 9, 2011, p. 2672-2713.
-
Johnston S. C., O'Meara E. S., Manolio T. A. et al. Cognitive impairment and decline are associated with carotid artery disease in patients without clinically evident cerebrovascular disease. Ann. Intern. Med., Vol. 140, Issue 4, 2004, p. 237-247.
-
Lee Y. H., Yeh S. J. Correlation of common carotid artery intima media thickness, intracranial arterial stenosis and post-stroke cognitive impairment. Acta Neurol. Taiwan, Vol. 16, Issue 4, 2007, p. 207-213.
-
Talelli P., Ellul J., Terzis G. et al. Common carotid artery intima media thickness and post-stroke cognitive impairment. J. Neurol Sci., Vol. 223, Issue 2, 2004, p. 129-134.
-
Sztriha L. K., Nemeth D., Sefcsik T. et al. Carotid stenosis and the cognitive function. J. Neurol. Sci., Vol. 283, Issue 1-2, 2009, p. 36-40.
-
Silvestrini M., Viticchi G., Falsetti L. et al. The role of carotid atherosclerosis in Alzheimer's disease progression. J. Alzheimers Dis., Vol. 25, Issue 4, 2011, p. 719-726.
-
Coll B., Feinstein S. B. Carotid intima-media thickness measurements: techniques and clinical relevance. Curr. Atheroscler. Rep., Vol. 10, Issue 5, 2008, p. 444-450.
-
Molinari F., Zeng G., Suri J. S. A state of the art review on intima-media thickness (IMT) measurement and wall segmentation techniques for carotid ultrasound. Comput. Methods Programs Biomed., Vol. 100, Issue 3, 2010, p. 201-221.
-
Lorenz M. W., Markus H. S., Bots M. L. et al. Prediction of clinical cardiovascular events with carotid intima-media thickness: a systematic review and meta-analysis. Circulation, Vol. 115, Issue 4, 2007, p. 459-467.
-
Bianchini E., Bozec E., Gemignani V. et al. Assessment of carotid stiffness and intima-media thickness from ultrasound data: comparison between two methods. J. Ultrasound Med., Vol. 29, Issue 8, 2010, p. 1169-1175.
-
Simon A., Megnien J. L., Chironi G. The value of carotid intima-media thickness for predicting cardiovascular risk. Arterioscler. Thromb. Vasc. Biol., Vol. 30, Issue 2, 2010, p. 182-185.
-
Sander K., Bickel H., Förstl H. et al. Carotid intima media thickness is independently associated with cognitive decline. The INVADE study. Int. J. Geriatr. Psychiatry, Vol. 25, Issue 4, 2010, p. 389-394.
-
Komulainen P., Kivipelto M., Lakka T. A. et al. Carotid intima-media thickness and cognitive function in elderly women: a population-based study. Neuroepidemiology, Vol. 28, Issue 4, 2007, p. 207-213.
-
Arntzen K. A., Mathiesen E. B. Subclinical carotid atherosclerosis and cognitive function. Acta Neurol. Scand. Suppl., Vol. 191, 2011, p. 18-22.
-
Knopman D., Boland L. L., Mosley T. et al. Atherosclerosis risk in communities (ARIC) study investigators. Cardiovascular risk factors and cognitive decline in middle-aged adults. Neurology, Vol. 56, Issue 1, 2001, p. 42-48.
-
Viswanathan A., Rocca W. A., Tzourio C. Vascular risk factors and dementia: how to move forward? Neurology, Vol. 72, Issue 4, 2009, p. 368-374.
-
Newman A. B., Fitzpatrick A. L., Lopez O. et al. Dementia and Alzheimer’s disease incidence in relationship to cardiovascular disease in the Cardiovascular Health Study cohort. J. Am. Geriatr. Soc., Vol. 53, 2005, p. 1101-1107.
-
Molinari F., Meiburger K. M., Saba L. et al. Ultrasound IMT measurement on a multi-ethnic and multi-institutional database: our review and experience using four fully automated and one semi-automated methods. Comput. Methods Programs Biomed., 2012 May 31, Epub ahead of print, PubMed PMID: 22658832.
-
Freire C. M., Ribeiro A. L., Barbosa F. B. et al. Comparison between automated and manual measurements of carotid intima-media thickness in clinical practice. Vasc. Health Risk Manag., Vol. 5, 2009, p. 811-817.
-
Rocha R., Campilho A., Silva J. et al. Segmentation of the carotid intima-media region in B-mode ultrasound images. Image and Vision Computing, Vol. 28, 2010, p. 614-625.
-
Seçil M., Altay C., Gülcü A. et al. Automated measurement of intima-media thickness of carotid arteries in ultrasonography by computer software. Diagn. Interv. Radiol., Vol. 11, Issue 2, 2005, p. 105-108.
-
Schreuder F. H., Graf M., Hameleers J. M. et al. Measurement of common carotid artery intima-media thickness in clinical practice: comparison of B-mode and RF-based technique. Ultras. Med., Vol. 30, Issue 5, 2009, p. 459-465.
-
Ciccone M. M., Scicchitano P., Zito A. et al. Correlation between coronary artery disease severity, left ventricular mass index and carotid intima media thickness, assessed by radio-frequency. Cardiovasc. Ultrasound, 2011 Nov. 16, p. 9-32.
-
Molinari F., Zeng G., Suri J. S. An integrated approach to computer-based automated tracing and its validation for 200 common carotid arterial wall ultrasound images: a new technique. J. Ultrasound Med., Vol. 29, Issue 3, 2010, p. 399-418.
-
Romero J. R., Beiser A., Seshadri S. et al. Carotid artery atherosclerosis, MRI indices of brain ischemia, aging, and cognitive impairment: the Framingham study. Stroke, Vol. 40, Issue 5, 2009, p. 1590-1596.
-
Arntzen K. A., Schirmer H., Johnsen S. H. et al. Carotid atherosclerosis predicts lower cognitive test results: a 7-year follow-up study of 4,371 stroke-free subjects – the Tromsø study. Cerebrovasc. Dis., Vol. 33, Issue 2, 2012, p. 159-165.
-
Dutra A. P. Cognitive function and carotid stenosis. Review of the literature. Dement. Neuropsychol., Vol. 6, Issue 3, 2012, p. 127-130.
-
Van Oijen M., De Jong F. J., Witteman J. C. et al. Atherosclerosis and risk for dementia. Ann. Neurol., Vol. 61, Issue 5, 2007, p. 403-410.
-
Faxon D. P., Creager M. A., Smith S. C. Jr. et al. American Heart Association. Atherosclerotic Vascular Disease Conference: Executive summary: Atherosclerotic Vascular Disease Conference Proceeding for healthcare professionals from a special writing group of the American Heart Association. Circulation, Vol. 109, Issue 21, 2004, p. 2595-2604.
